A potential new biomarker for Alzheimer’s
Alzheimer’s is considered a disease of old age, with most people being diagnosed after 65. But the condition actually begins developing out of sight many years before any symptoms emerge. Tiny proteins, known as amyloid-beta peptides, clump together in the brain to form plaques. These plaques lead to inflammation and eventually cause neuronal cell death.
Interplay of proteins in the brain reveals disease mechanism
We’re lacking good diagnostic markers that would allow us to reliably detect the disease at an early stage or make predictions about its course.
Exactly what triggers these pathological changes is still unclear. “We’re lacking good diagnostic markers that would allow us to reliably detect the disease at an early stage or make predictions about its course,” says Professor Erich Wanker, head of the Proteomics and Molecular Mechanisms of Neurodegenerative Diseases Lab at the Max Delbrück Center. Wanker and his team are studying brains with Alzheimer’s disease to understand their proteome – the interplay between all the proteins involved in the onset and course of the disease. Writing in “Genome Medicine”, the researchers now report on a new actor in the pathological process. Their discovery will help scientists understand the mechanisms underlying Alzheimer’s and could also serve as a marker for improved diagnostics.
To analyze changes in the proteome, Wanker’s team studies genetically modified mice. The mice have five mutations that occur in people with familial Alzheimer’s disease. The amyloid-beta plaques develop in the mice’s brains and the animals show typical symptoms, such as dementia.
New perspectives for a better understanding of Alzheimer’s
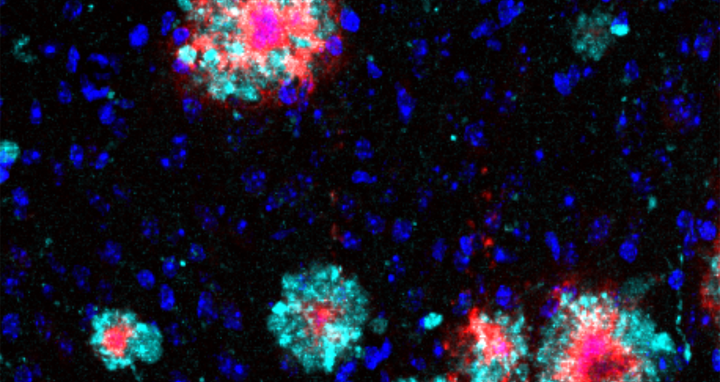
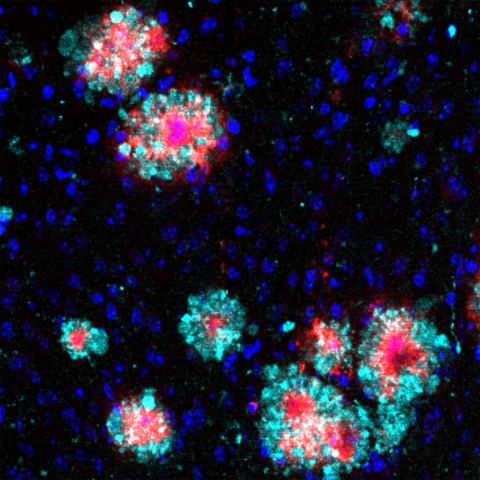
Brain section of an Alzheimer’s mouse: The Arl8b protein (turquoise) shows an accumulation around the amyloid‑b aggregates (red). Cell nuclei are stained blue. The section was analysed with immunofluorescence.
“During our analyses, we noticed that a protein called Arl8b was building up in mouse brains, along with the amyloid-beta plaques,“ says Annett Böddrich, lead author of the paper. The researchers also found accumulations of the protein in brain samples from Alzheimer’s patients.
Arl8b is associated with lysosomes, cell organelles that are involved in degrading the protein clumps. A different team of researchers recently made an interesting discovery in the nematode worm: increasing Arl8b production can degrade the plaques, which reduces the damage to nerve cells. Closer study of Arl8b could be the key to better understanding Alzheimer’s disease – and could provide a new target for therapies.
Interesting candidate for a diagnostic marker
But there’s more: “We can show that Alzheimer’s patients have significantly more Arl8b in their cerebrospinal fluid than healthy controls,” says Böddrich. Unlike brain tissue, cerebrospinal fluid is easily accessible for diagnostic studies. “This means Arl8b is an interesting candidate for a diagnostic marker,” she says.
However, the study only looked at a small group of Alzheimer’s patients, so expectations should be kept in check: “It’s too early to hope for a diagnostic test,” says Wanker. Nevertheless, he is optimistic: “Our work shows that proteomic research can provide crucial information for identifying disease mechanisms and markers, and thereby move research forward. Also, this doesn’t just apply to Alzheimer’s; it’s also relevant to other complex neurodegenerative diseases such as Parkinson’s and Huntington’s.”
Text: Stefanie Reinberger
Further information
Literatur
Annett Boeddrich et al. (2023): “A proteomics analysis of 5xFAD mouse brain regions reveals the lysosome associated protein Arl8b as a candidate biomarker for Alzheimer’s disease,” Genome Medicine, DOI: 10.1186/s13073-023 – 01206‑2
Downloads
Brain section of an Alzheimer’s mouse: The Arl8b protein (turquoise) shows an accumulation around the amyloid‑b aggregates (red). Cell nuclei are stained blue. The section was analysed with immunofluorescence. Image: AG Wanker, Max Delbrück Center
Press contacts
Professor Erich E. Wanker
Head of the lab „Proteomics and Molecular Mechanisms of Neurodegenerative Diseases”
Max Delbrück Center
+49 30 9406 – 2157
ewanker@mdc-berlin.de
Christina Anders
Editor, Communications
Max Delbrück Center
+49 30 9406 – 2118
christina.anders@mdc-berlin.de or presse@mdc-berlin.de
- Max Delbrück Center
-
The Max Delbrück Center for Molecular Medicine in the Helmholtz Association (Max Delbrück Center) is one of the world’s leading biomedical research institutions. Max Delbrück, a Berlin native, was a Nobel laureate and one of the founders of molecular biology. At the locations in Berlin-Buch and Mitte, researchers from some 70 countries study human biology – investigating the foundations of life from its most elementary building blocks to systems-wide mechanisms. By understanding what regulates or disrupts the dynamic equilibrium of a cell, an organ, or the entire body, we can prevent diseases, diagnose them earlier, and stop their progression with tailored therapies. Patients should be able to benefit as soon as possible from basic research discoveries. This is why the Max Delbrück Center supports spin-off creation and participates in collaborative networks. It works in close partnership with Charité – Universitätsmedizin Berlin in the jointly-run Experimental and Clinical Research Center (ECRC), the Berlin Institute of Health (BIH) at Charité, and the German Center for Cardiovascular Research (DZHK). Founded in 1992, the Max Delbrück Center today employs 1,800 people and is 90 percent funded by the German federal government and 10 percent by the State of Berlin.