Strong change of course for muscle research
Cells from which new muscles arise have enormous potential for developing gene therapies to treat muscle atrophy.
Anyone who climbs the 285 steps to the viewing platform of Berlin’s Siegessäule, or Victory Column, will probably have quite a few sore muscles the next day. Out-of-the-ordinary activities such as climbing lots of steps or even normal exercise can put significant strain on muscles. Such activities cause tiny tears in the muscle fibers, which the body then repairs on its own.
Even when injuries occur, the muscles activate an endogenous regeneration program: A reserve supply of muscle stem cells, known as satellite cells, reside around the muscle fibers and are essential for the repair of damaged muscle cells. These satellite cells produce new muscle fibers in a process which results in muscle regeneration. People maintain this ability well into old age. Researchers are particularly interested in these cells since they could provide targets for new therapeutic approaches for people with muscle diseases.
An overrated protein
Researchers previously assumed that a certain protein – the transcription factor PAX7 – plays a key role in muscle regeneration. “Cells from which new muscles arise have enormous potential for developing gene therapies to treat muscle atrophy. And PAX7 is actually considered a characteristic property of muscle-building satellite cells,” says Prof. Simone Spuler.
The scientist and physician is a research group leader at the Experimental and Clinical Research Center (ECRC), a joint institution of the Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC) and Charité – Universitätsmedizin Berlin, and heads the Myology Group at the MDC. Her team has now reported in the journal Nature Communications that it’s possible for muscles to grow and regenerate without PAX7. The study characterized a previously unknown subtype of satellite cells that could play an important role in the future development of gene therapies from muscle stem cells.
Our findings lead us down a new path.
“The findings will certainly surprise many researchers in the field,” says Dr. Andreas Marg, a senior scientist in Spuler’s lab and the lead author of the study. He himself was initially guided by the assumption that the transcription factor was crucial for muscle growth. “I previously focused my research on PAX7-positive cells. Our findings lead us down a new path.”
New muscles despite a mutation
Patient selfie.
The research team owes the discovery to a young girl: Lavin has suffered from a genetic form of muscular dystrophy since birth and is the protagonist in the study. Lavin has all the muscles of a healthy person, but each of her muscles is very small. The musculature along her spine is particularly affected by the disease. Lavin’s arms and legs are strong, but she suffers breathing problems and has difficulty bending forward and holding her head up.
Gene analysis shows that the gene for PAX7 is damaged in Lavin; her cells can’t produce this protein. The University Hospital Munich discovered this in 2017. Soon thereafter, Spuler and Marg learned of this extremely rare mutation – one that had not been described before. Lavin traveled with her parents to the Berlin-Buch campus, where the scientists took a sample of her muscle tissue. Marg used a new procedure to filter out Lavin’s satellite cells and then implanted them in mice. He observed that new muscle fibers grew in the mice from Lavin’s cells – despite the absence of PAX7.
We could perhaps develop a gene therapy for Lavin using the CRISPR-Cas9 gene-editing tool.
Spuler presumes that PAX7 is not equally important for every cell. This would explain why Lavin can walk and climb relatively well, but has hardly any strength in her diaphragm, which causes the breathing problems. “We could perhaps develop a gene therapy for Lavin using the CRISPR-Cas9 gene-editing tool,” says Spuler. “However, to repair the defective gene, CRISPR-Cas9 would have to specifically target the cells of the axial musculature, and that is not yet possible.” But Spuler’s lab is working intensively to figure out how to repair defective genes in muscle cells. For Lavin and her family, this research offers a small glimmer of hope that a suitable therapy will be found.
A new subtype of muscle stem cells
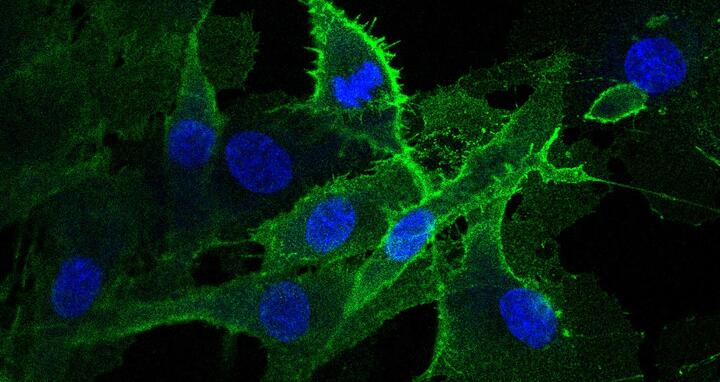
The muscle cell population of Lavin shows a very strong expression of the surface protein CLEC14A (green). Image: Spuler Lab, MDC
Marg and Spuler collaborated on the study with many colleagues at the MDC and with scientists from institutions abroad. Prof. Nikolaus Rajewsky’s research group at the Berlin Institute for Medical Systems Biology (BIMSB) compared Lavin’s cells with those donated by healthy people. Single-cell analysis, which looks at the activity of each cell individually, revealed a previously unknown cell population. In around 20 percent of the donors, the majority of the activated satellite cells also don’t produce any PAX7, even though the genetic information is present in the cells. The team instead discovered something else in those cells in which the transcription factor was missing: CLEC14A, a protein that is found in many blood vessel cells. This very protein was highly expressed in Lavin’s muscle stem cells.
The new study describes a previously unknown subtype of satellite cells. First, the researchers identified these cells in the stem cell niche, which is where the satellite cells reside. Second, PAX7 is not present in these cells. Third, other characteristic proteins such as CLEC14A are present instead. And fourth, new muscle fibers can be derived from this cell population.
Up to now, only cells with PAX7 have been considered as targets for gene therapy research involving satellite cells. The new study shows that the subtype discovered should also play a role in therapeutic development.
Further information
Fotos zum Download
- The muscle cell population of Lavin shows a very strong expression of the surface protein CLEC14A (green). Image: Spuler Lab, MDC
- Patient selfie Image: privat
Literature
Marg, et al. (2019): “Human muscle-derived CLEC14A-positive cells regenerate muscle independent of PAX7”, Nature Communications, DOI: 10.1038/s41467-019 – 13650‑z
Press contacts
Prof. Simone Spuler
Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC) and Charité – Universitätsmedizin Berlin
Head of the Myology Research Group
+49 (0)30 450 5405 01 or +49 (0)30 450 5405 04
simone.spuler@mdc-berlin.de or simone.spuler@charite.de
Christina Anders
Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC)
Editor, Communications Department
+49 (0)30 9406 2118christina.anders@mdc-berlin.de or presse@mdc-berlin.de
- The Max Delbrück Center for Molecular Medicine (MDC)
-
The Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC) is one of the world’s leading biomedical research institutions. Max Delbrück, a Berlin native, was a Nobel laureate and one of the founders of molecular biology. At the MDC’s locations in Berlin-Buch and Mitte, researchers from some 60 countries analyze the human system – investigating the biological foundations of life from its most elementary building blocks to systems-wide mechanisms. By understanding what regulates or disrupts the dynamic equilibrium in a cell, an organ, or the entire body, we can prevent diseases, diagnose them earlier, and stop their progression with tailored therapies. Patients should benefit as soon as possible from basic research discoveries. The MDC therefore supports spin-off creation and participates in collaborative networks. It works in close partnership with Charité – Universitätsmedizin Berlin in the jointly run Experimental and Clinical Research Center (ECRC), the Berlin Institute of Health (BIH) at Charité, and the German Center for Cardiovascular Research (DZHK). Founded in 1992, the MDC today employs 1,600 people and is funded 90 percent by the German federal government and 10 percent by the State of Berlin.